Previous Page | Next Page | Table of Contents | Diagram Center Home
Basic and Amphoteric Hydroxides
Source: Boundless. “Basic and Amphoteric Hydroxides.” Boundless Chemistry. Boundless, 19 Nov. 2014. Retrieved 10 Feb. 2015 from https://www.boundless.com/chemistry/textbooks/boundless-chemistry-textbook/acids-and-bases-15/acid-base-properties-of-oxides-114/basic-and-amphoteric-hydroxides-469-6403/
This page, from an introductory postsecondary textbook, includes chemical equations and 2D molecular structure diagrams called Lewis structures, which are common in chemistry textbooks. For an example of 3D models of molecular and atomic structures, which are also common in this discipline, please see Sample 6: Carbon Atom.
Making This Image Accessible
This image can be made accessible using MathML, image descriptions, or by creating a tactile graphic. Lewis structures can also be made accessible to braille readers through the use of chemical notation braille code. Further, other possibilities include the use of Scalable Vector Graphics or the inclusion of chemical file formats, which may be used with accessible molecular viewing software. The choice of modality varies depending on factors such as the information to be conveyed, grade level, student knowledge and experience, and the image itself.
How an image description is displayed depends on the reading tool being used. In most mainstream reading tools, the description will not be displayed at all, but in tools designed for accessibility (or mainstream tools with accessible features enabled), the description could be displayed below the image, voiced as part of the content, or both.
MathML
Mathematical Markup Language (MathML) is most commonly used in creating mathematics equations, but it works equally well to create accessible chemical equations. Using MathML provides flexibility in the speech or braille output generated by assistive technology when supported. For instance, intelligent speech systems can render the formula HCl as “hydrochloric acid,” which is the standard International Union of Pure and Applied Chemistry (IUPAC) nomenclature for this compound. In supported systems, MathML may also allow an appropriate braille output.
For example, the code used to create this equation:
HCl(aq)+Al(OH)3(aq)→AlCl3(aq)+3H2O(l)
looks like:
<math>
<mrow>
<mi mathvariant="normal">H</mi>
<mi mathvariant="normal">Cl</mi>
<mo stretchy="false">(</mo>
<mi>a</mi>
<mi>q</mi>
<mo stretchy="false">)</mo>
<mo>+</mo>
<mi mathvariant="normal">Al</mi>
<msub>
<mrow>
<mo stretchy="false">(</mo>
<mi mathvariant="normal">O</mi>
<mi mathvariant="normal">H</mi>
<mo stretchy="false">)</mo>
</mrow>
<mn>3</mn>
</msub>
<mo stretchy="false">(</mo>
<mi>a</mi>
<mi>q</mi>
<mo stretchy="false">)</mo>
<mo>→</mo>
<mi mathvariant="normal">Al</mi>
<msub>
<mrow>
<mi mathvariant="normal">Cl</mi>
</mrow>
<mn>3</mn>
</msub>
<mo stretchy="false">(</mo>
<mi>a</mi>
<mi>q</mi>
<mo stretchy="false">)</mo>
<mo>+</mo>
<mn>3</mn>
<msub>
<mi mathvariant="normal">H</mi>
<mn>2</mn>
</msub>
<mi mathvariant="normal">O</mi>
<mo stretchy="false">(</mo>
<mi>l</mi>
<mo stretchy="false">)</mo>
</mrow>
</math>Text or Audio Description
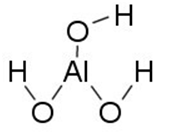
It is possible to create comprehensible text descriptions to include in digital formats. For the image included below, the description is short enough that only alt text is needed. Using the proper IUPAC naming convention for elements and compounds, is typically the best approach.
“Aluminum bonded to 3 Hydroxyl groups.”
Previous Page | Next Page | Table of Contents | Diagram Center Home