<< Previous section: Art, Photos & Cartoons | Main Table of Contents | Next section: Diagrams (Illustrated) >>
B. Chemistry:
1. Chemical Element
Guidelines:
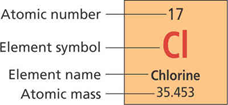
- When describing one element from the periodic table, write out the four property labels in narrative form as is listed in the periodic table, from top to bottom. See Description 1.
- If the information required is about the properties of the element, change the order of the information to put the chemical name first, then list its properties. See Description 2
- When listing two to three elements, use a bullet format. When listing three or more elements, use a table format for easier navigability. The property labels of the element would be listed across the top as a header.
- When a screen reader reads an element symbol, it may not pronounce that symbol correctly. If that is a concern, an option to prevent that from happening is to use all caps and put a space or punctuation in between the letters. For example, the symbol for Chlorine could be written as C L, or C, L.
Description 1:
A diagram shows properties for the element Chlorine from the periodic table of elements. Atomic number, 17. Element symbol, Cl. Element name, Chlorine. Atomic mass, 35.453.Description 2:
An element from the periodic table. Element name, Chlorine. Atomic number, 17. Element symbol, Cl. Atomic mass, 35.453.This image in context:
- This image appears at the beginning of the section.
- The caption explains that there are four pieces of information about an element on the periodic table of elements.
- In context, Description 1 would work best.
<< Previous: Section A (Art) |Return to main table of contents | Return to top of page | Next: Section C (Diagrams: Illustrated) >>

